THEORY:
All the molecules of a liquid on the surface experience a resultant downward pull. This resultant downward pull or force of attraction which acts perpendicularly to any line of 1 cm length on the surface of the liquid is called the surface tension of the liquid. It is denoted by the symbol γ, and its unit is dynes per cm in c.g.s units, and newton per meter in S.I units. These two units are related as
1 Nm-1 = 1000 dynes cm-1
The surface tension of the given liquid can be calculated by using the relation.
γ1/γ2 = n2d1 / n1d2 …………(i)
Where, γ1 and γ2 = surface tension of the liquid and water respectively.
n1 and n₂ = number of drops of liquid and water falling down for the same volume V from the capillary of the stalagmometer.
d1 and d₂ = density of the liquid and water respectively.
APPARATUS :
1) Stalagmometer
2) Pyknometer
3) Beaker
4) Balance
5) Thermometer
PROCEDURE:
i) Clean the stalag-mometer and the pycnometer (or the specific gravity bottle) first with chromic acid and then with distilled water. Dry them in an air oven at about 120°C.
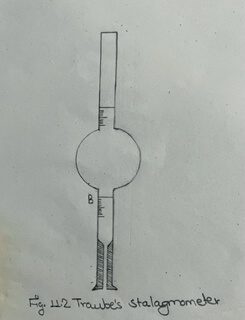
ii) Clamp the stalagmometer vertically, and attach a rubber tubing with a screw pinch cock at the upper end.
iii) Dip the lower end of the stalagmometer in a beaker containing the liquid, and from the upper end suck till the liquid rises above the upper mark of the stalagmometer. Adjust the level of the liquid just to the upper mark. Now adjust the screw of the pinch cock so that the liquid falls down at the rate of one drop in every three seconds. Refill the stalagmometer with the liquid, and count the number of drops falling when the volume of the liquid flows from the upper to the lower mark. Repeat the process at least three times.
vi) Clean the stalagmometer, and repeat the whole process for distilled water.
v) Determine the weight of an empty pycnometer (or specific gravity bottle). Next, fill the pycnometer with distilled water and take its weight. Now empty the pycnometer and dry it in the air oven. Finally, take the weight of the pycnometer filled with the liquid under investigation.
RESULTS:
Room temperature at the time of experiment = t°C
Surface tension of water at t°C= ……… dyne cm-1
| No.of obs. | No. of dropsof liquid (n1) | Mean n1 | No. of drops ofwater (n₂) | Mean (n₂) |
| 1 | ||||
| 2 | ||||
| 3 |
Wt of empty pycnometer = a gm
Wt of pycnometer + water = b gm
Wt of pycnometer + liquid = c gm
Therefore, Wt of water in the pycnometer = (b – a) gm
and wt of liquid in the pycnometer = (c – a) gm
therefore, d1 / d₂ = (c-a) / (b-a)
Calculate the surface tension of liquid by the relation
γ1 = n2 d1 / n1 d₂ x γ2 dyne cm-2
